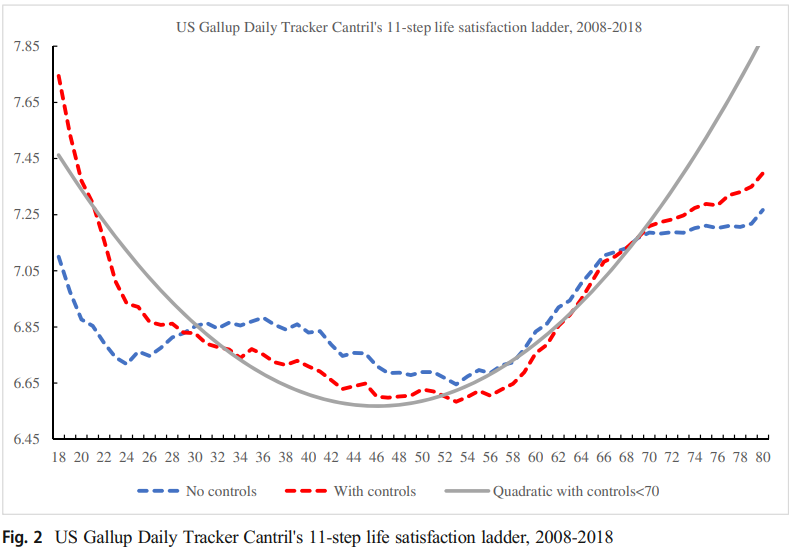
Zacks Small Cap Analysis – LGVN Secures Operations By Essential Take a look at – Cyber Tech
By Brad Sorensen, CFA
READ THE FULL LGVN RESEARCH REPORT
Longeveron (NASDAQ:LGVN) is a clinical-stage biotechnology firm that has positioned itself within the regenerative medication house with a doubtlessly life-changing therapeutic candidate. Its flagship product, laromestrocel, is an allogeneic medicinal-signaling cell (MSC) remedy derived from younger wholesome grownup donors and manufactured underneath present good manufacturing follow. The science behind laromestrocel rests on the concept these MSCs might present anti-inflammatory, pro-vascular, and regenerative help in tissues which are broken or underneath stress. The corporate behind these thrilling developments is at an inflection level in our view, with an excellent alternative to realize lots of the targets mentioned within the close to future.
To additional that purpose, Longeveron Inc. just lately introduced a brand new non-public placement financing that would present the corporate with as much as $30 million in whole gross proceeds, structured in two tranches and led by institutional traders.
The primary tranche is anticipated to generate roughly $15 million at closing, with participation from traders together with Coastlands Capital and Janus Henderson Traders, whereas H.C. Wainwright is serving because the unique placement agent. The financing is priced at $0.52 per share, roughly at-the-market underneath Nasdaq guidelines. Within the preliminary closing, the corporate will situation about 6.0 million shares of Class A standard inventory, together with Sequence A non-voting convertible most popular shares which are convertible into roughly 22.8 million further frequent shares on the similar $0.52 conversion worth.
The settlement additionally features a second potential tranche of as much as $15 million, which might be funded if sure situations are met. These situations are tied primarily to scientific milestones associated to the Part 2b ELPIS II trial in hypoplastic left coronary heart syndrome (HLHS) and the corporate’s share worth efficiency.
A further component of the deal provides traders an curiosity in 50% of proceeds from any future sale of a Uncommon Pediatric Illness Precedence Evaluate Voucher, ought to the corporate obtain one from the FDA in reference to its laromestrocel remedy for HLHS.
Administration acknowledged that the proceeds, mixed with present money, are anticipated to fund operations into the fourth quarter of 2026, carrying the corporate past the anticipated top-line information readout from the pivotal ELPIS II trial within the third quarter of 2026. Funds will primarily help scientific growth of the corporate’s stem-cell remedy laromestrocel, together with working capital and basic company functions.
Total, the financing supplies LGVN with near-term capital to advance its lead scientific program whereas tying a portion of the funding to imminent scientific milestones, although it additionally introduces potential dilution by means of convertible securities tied to the $0.52 conversion worth.
We need to remind traders why this funding is so necessary because of the progress made on treating HLHS, which is a uncommon and life-threatening congenital coronary heart defect during which the left ventricle is severely underdeveloped or absent, which means that the best ventricle should be tailored to deal with systemic circulation. Even with the usual of care—usually a sequence of three reconstructive surgical procedures over the primary few years of life—survival into adolescence is simply round 50–60%.
Longeveron’s strategy has been to manage laromestrocel instantly into the best ventricle (or the myocardium of the best ventricle) through the second stage of surgical procedure (the “Glenn” process, usually at round 4 months of age). Firm scientists imagine that by bettering the operate of the systemic proper ventricle—by means of regenerative mechanisms—the remedy ought to enhance transplant-free survival and long-term outcomes for these infants.
The corporate reported Part 1 outcomes (known as ELPIS I) in ten infants. In that examine, laromestrocel was nicely tolerated: there have been no main adversarial cardiovascular occasions or infections associated to remedy by means of one yr, assembly the first security endpoint. Much more putting, long-term follow-up confirmed 100 % transplant-free survival as much as 5 years in these sufferers, in comparison with historic controls (roughly 83 % at 5 years, with ~5 % requiring transplant) within the comparable inhabitants.
Constructing on that, Longeveron is now conducting a pivotal Part 2b trial (ELPIS II-mentioned above). The trial is designed to match Laromestrocel plus normal surgical procedure versus normal surgical procedure alone, with endpoints together with survival at 12 months, size of hospitalization and alter in proper ventricular ejection fraction between baseline and 12 months. Full enrollment was achieved in June 2025, with topline outcomes, as talked about above, anticipated within the third quarter of 2026 after a 12-month follow-up.
On the regulatory entrance, this system has very sturdy designations. The U.S. Meals and Drug Administration has authorised three particular designations for laromestrocel within the HLHS indication: Uncommon Pediatric Illness (RPD) designation, Orphan Drug Designation (ODD), and Quick Observe designation. Importantly, following a Kind C assembly with FDA in September 2024, the company confirmed that the ELPIS II trial may function a pivotal examine—and if constructive, may kind the premise for a Biologics License Utility (BLA) submission for full conventional approval.
Abstract
We proceed to imagine that Longeveron is an thrilling clinical-stage firm, and traders aren’t appropriately appreciating the game-changing potential laromestrocel might be able to have on a number of severe medical situations. Because of the prudent choices made by administration, we imagine laromestrocel will finally have a considerable affect on the well being conditions of 1000’s of sufferers. We imagine the inventory continues to be underpriced as traders aren’t appreciating the potential of laromestrocel and encourage traders to check out LGVN.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to obtain our articles and stories emailed on to you every morning. Please go to our web site for added data on Zacks SCR.
DISCLOSURE: Zacks SCR has obtained compensation from the issuer instantly, from an funding supervisor, or from an investor relations consulting agency, engaged by the issuer, for offering analysis protection for a interval of a minimum of one yr. Analysis articles, as seen right here, are a part of the service Zacks SCR supplies and Zacks SCR receives funds totaling a most charge of as much as $50,000 yearly for these companies offered to or concerning the issuer. Full Disclaimer HERE.